Chiglitazar Sodium (trade name: Bilessglu®) is a novel molecular entity drug with a unique mechanism discovered exclusively by the company. It is the world's first PPAR pan-agonist and belongs to the insulin sensitizer class. Chiglitazar Sodium is an achievement of China's National 863 Program and the "Major New Drug Innovation" initiative.
By moderately activating all three PPAR receptors, Chiglitazar Sodium achieves dynamic balance in glucose, lipid, and energy metabolism. It improves metabolic diseases through multiple mechanisms, including glucose-lowering, insulin sensitization, lipid regulation, anti-inflammatory effects, and anti-fibrotic actions. Multiple clinical studies have demonstrated the efficacy and safety of Chiglitazar Sodium in treating type 2 diabetes and fatty liver disease.
Chiglitazar Sodium has been approved for diabetes indications and included in the National Reimbursement Drug List, and is incorporated into authoritative guidelines/consensus such as the "Chinese Guidelines for the Prevention and Treatment of Diabetes (2024)", "National guidelines for the prevention and control of diabetes in primary care (2025)", and "Expert Consensus on Oral Hypoglycemic Drug Combination Therapy for Adults with Type 2 Diabetes (2025)". In March 2026, the company received the Drug Clinical Trial Approval Notice for Chiglitazar Sodium/Metformin Sustained-Release Tablets (combination product) and will conduct clinical studies as required for marketing registration. Chiglitazar Sodium is continuously accumulating clinical evidence in metabolic fields such as diabetes and metabolic dysfunction-associated fatty liver disease, and is expected to become a foundational drug for the comprehensive treatment of metabolic diseases.
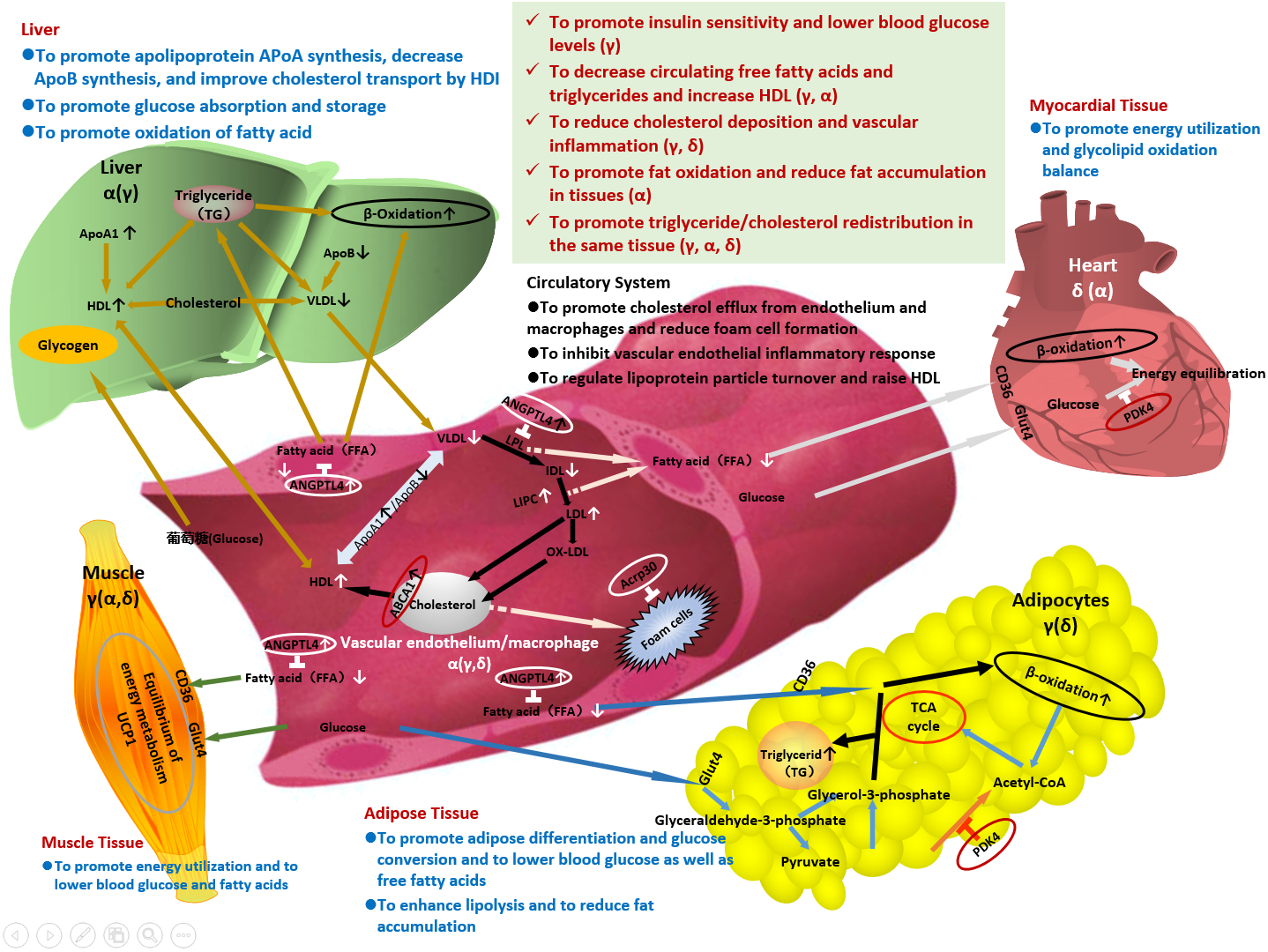
Figure Potential Mechanisms of Action of Chiglitazar Sodium
Commercialization Status in China:
- In October 2021, Chiglitazar Sodium was approved for the treatment of type 2 diabetes (T2DM).
- In March 2023, Chiglitazar Sodium was included in the National Medical Insurance Drug List.
- In July 2024, Chiglitazar Sodium in combination with metformin was approved for the treatment of patients with type 2 diabetes mellitus inadequately controlled by metformin alone, providing a new option for combination therapy in type 2 diabetes patients.
- In November 2024, Chiglitazar Sodium was renewed at the original price in the National Medical Insurance Drug List.
Continuously expanding clinical evidence:
- In March 2024, a randomized, double-blind, placebo-controlled phase II clinical trial evaluating Chiglitazar Sodium monotherapy in the treatment of metabolic dysfunction-associated steatohepatitis (MASH) achieved its primary efficacy endpoints with favorable efficacy.
- In November 2024, the results of this study were presented at an oral presentation at the 75th Annual Meeting of the American Association for the Study of Liver Diseases (AASLD), delivered by Professor You Hong from Beijing Friendship Hospital, Capital Medical University, during the conference.
- In March 2025, at the Annual Meeting of the Asian Pacific Association for the Study of the Liver, Professor Zang Shufei from the Department of Endocrinology at Shanghai Fifth People's Hospital delivered an oral presentation on a new clinical study of Chiglitazar Sodium for the treatment of type 2 diabetes complicated with metabolic dysfunction-associated steatohepatitis. The study revealed that the pan-PPAR agonist Chiglitazar Sodium (Bilessglu®) 48 mg demonstrated favorable efficacy and safety in the treatment of high-risk populations with T2DM and MASH, offering new insights and options for clinical treatment.
- In June 2025, a real-world study on the effects of Chiglitazar Sodium on type 2 diabetes complicated with metabolic dysfunction-associated fatty liver disease was published in Diabetes & Metabolism, the official journal of the French Society for the Study of Diabetes (SFD). The study showed that Chiglitazar Sodium significantly reduced the controlled attenuation parameter (CAP) value, a key indicator of liver fatty in patients with metabolic dysfunction-associated steatotic liver disease and Type 2 diabetes mellitus, indicating its potential as a dual therapy for co-management of diabetes and liver disease.
- In August 2025, the phase II clinical trial results of Chiglitazar Sodium monotherapy for metabolic dysfunction-associated steatohepatitis were published in Hepatology, a top-tier international journal in the field of liver diseases. After 18 weeks of treatment, patients showed a 40% reduction in liver fat content, with more than 70% achieving normalization of liver enzyme levels.
- In September 2025, the "PRECISE Study" of Chiglitazar Sodium was initiated to promote prediabetes intervention.
- In September 2025, a landmark study on PPARs was presented as an oral report at the Annual Meeting of the European Association for the Study of Diabetes (EASD 2025), providing new evidence for the treatment of diabetic foot ulcers.

