Chiauranib is a new molecular entity exclusively discovered by the company with global patent protection, featuring a novel mechanism as a triple-pathway tumor-targeting inhibitor. Chiauranib selectively inhibits protein kinase targets related to tumorigenesis and tumor development, including Aurora B, CSF1R, and VEGFR/PDGFR/c-Kit. It exerts multi-directional and broad-spectrum anti-tumor effects by inhibiting tumor cell proliferation, suppressing tumor angiogenesis, and regulating the tumor immune microenvironment, demonstrating favorable efficacy and safety in clinical trials of monotherapy for end-line tumor treatment.
With its highly selective inhibition of Aurora B/VEGFR/PDGFR/c-Kit/CSF1R targets, Chiauranib achieves comprehensive anti-tumor efficacy through three active mechanisms: inhibiting tumor cell mitosis/genomic stability, suppressing tumor angiogenesis, and modulating the tumor immune microenvironment. This multi-pathway mechanism results in superior pre-clinical pharmacodynamic activity compared to drugs with similar mechanisms and exhibits a favorable safety profile.
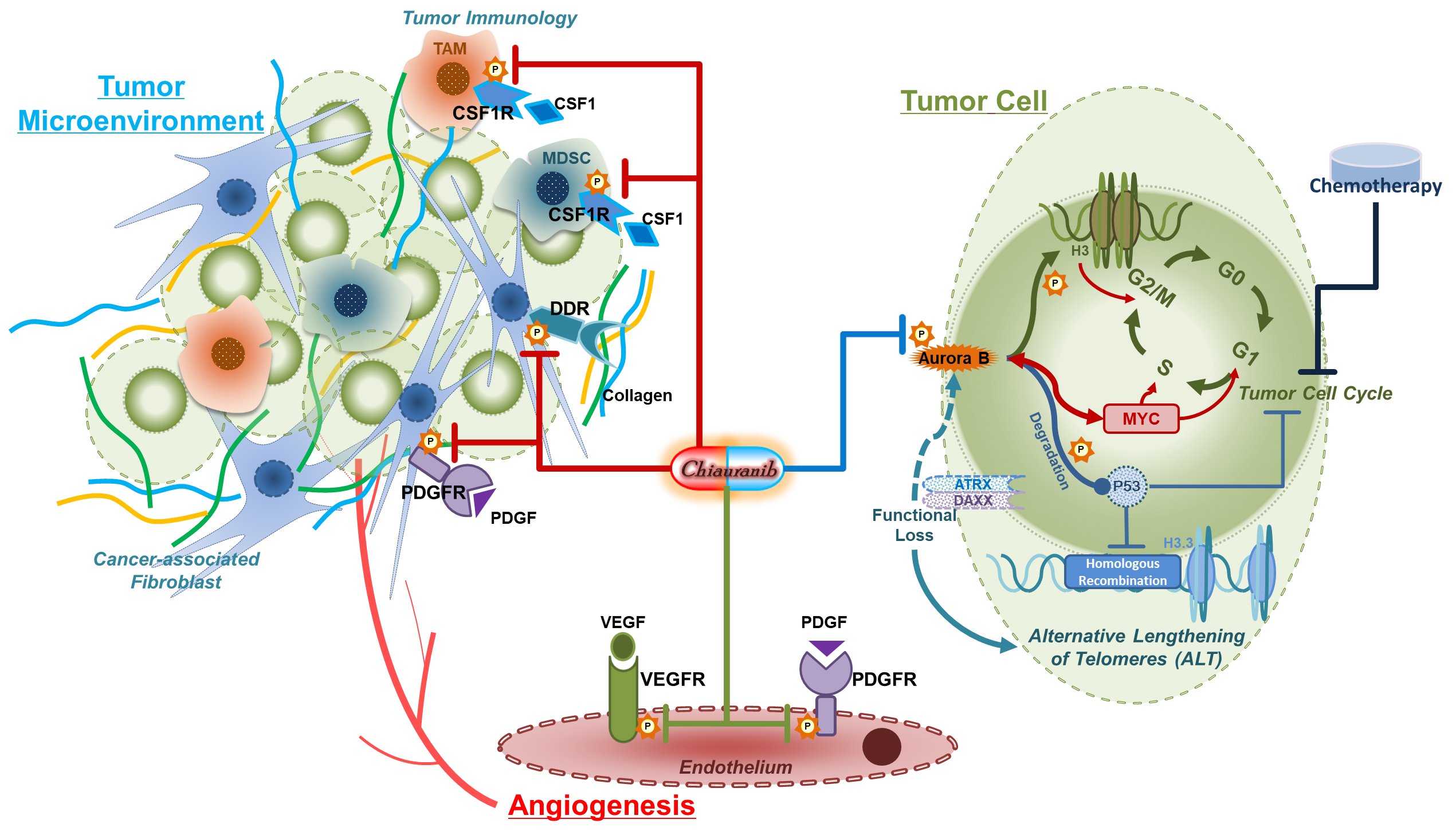
Figure Schematic Diagram of the Triple-Pathway Anti-Tumor Mechanism of Chiauranib
Global Pipeline of Investigational Indications:
- The Phase II clinical study of Chiauranib in combination with albumin-bound paclitaxel and gemcitabine as first-line treatment for patients with pancreatic ductal adenocarcinoma has completed enrollment. At a median follow-up of 9.6 months, the ORR (objective response rate) for the Chiauranib combination regimen reached 50%, with approximately half of the patients still on continuous treatment. The mPFS (median progression-free survival) was 9.1 months (95% CI: 6.2-10.9), and OS (overall survival) data were not yet mature. The efficacy of this study was significantly superior to historical chemotherapy data, with favorable safety and tolerability in patients.
- In January 2026, the Phase III clinical study of Chiauranib in combination with toripalimab, albumin-bound paclitaxel, and gemcitabine as first-line treatment for patients with metastatic pancreatic ductal adenocarcinoma was approved by the NMPA.
- The Phase III, randomized, double-blind, controlled, multicenter clinical study of Chiauranib in combination with Paclitaxel for the treatment of patients with platinum-refractory or platinum-resistant relapsed ovarian cancer is ongoing.
- The Phase Ib/II clinical study of Chiauranib in the U.S. is currently escalating the dose to 110mg, with no dose-limiting toxicities (DLT) observed so far.