Chidamide (trade name: Epidaza®) is a new molecular entity drug exclusively discovered by the company with a novel mechanism. It is the world's first subtype-selective histone deacetylase (HDAC) inhibitor and the first oral therapeutic drug approved for the treatment of peripheral T-cell lymphoma and advanced hormone receptor-positive breast cancer, belonging to the class of epigenetic modulators.
Chidamide acts on epigenetics-related targets—histone deacetylases (subtypes 1, 2, and 3 of Class I and subtype 10 of Class IIb). Histone deacetylases (HDACs) are a class of proteases that play a crucial role in chromosomal structural modification and gene expression regulation. As an HDAC inhibitor, Chidamide exerts its effects by inhibiting the biological activity of HDACs, thereby inducing changes in the gene expression of multiple signaling pathways involved in tumorigenesis (i.e., epigenetic changes).
Specifically, the general mechanisms of action of Chidamide primarily include: ① Directly inhibiting the tumor cell cycle and inducing apoptosis; ② Inducing and activating natural killer (NK) cell- and antigen-specific cytotoxic T lymphocyte (CTL)-mediated tumor killing effects; ③ Inhibiting phenotypic transformation of tumor cells and the pro-drug resistance/pro-metastasis activity in the tumor microenvironment.
Chidamide has been included in the National Reimbursement Drug List as a Class B drug in China, with approved indications for peripheral T-cell lymphoma (PTCL), breast cancer, and diffuse large B-cell lymphoma (DLBCL). In Japan, Chidamide has been approved for adult T-cell leukemia (ATL) and PTCL, while in Taiwan, China, it has been approved for breast cancer. Currently, the company and its partners are exploring the application of Chidamide in combination with tumor immunotherapy worldwide and developing next-generation HDACi+IO drugs.
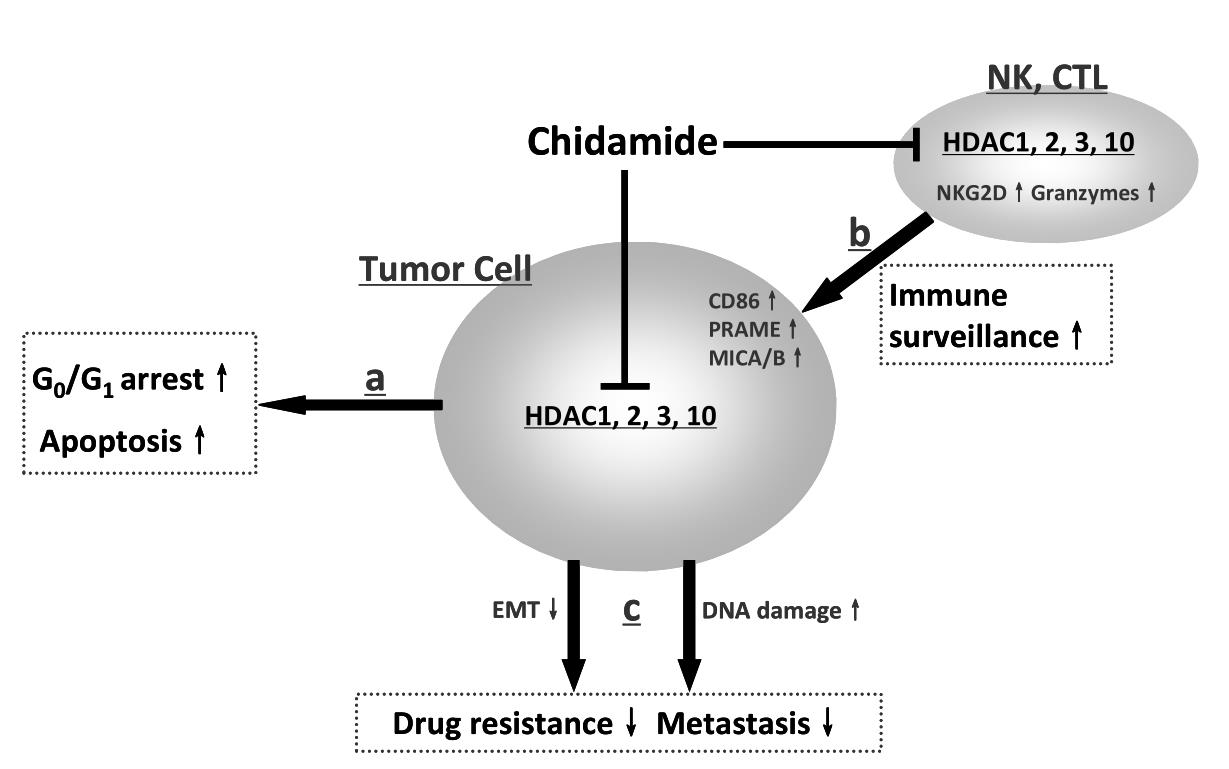
Figure: Molecular Anti-tumor Mechanisms of Action of Chidamide (Epidaza)
Commercialization Status:
- In December 2014, it was approved in China for the treatment of patients with relapsed or refractory peripheral T-cell lymphoma (PTCL) who had received at least one prior systemic chemotherapy, filling a gap in this treatment area in the country.
- In July 2017, it was included in the National Medical Insurance Drug List, benefiting more patients.
- In November 2019, it was approved in China in combination with an aromatase inhibitor for the treatment of postmenopausal patients with locally advanced or metastatic breast cancer that is hormone receptor-positive, human epidermal growth factor receptor 2 (HER2)-negative, and has relapsed or progressed after endocrine therapy. It was first confirmed that HDAC inhibitors in combination with other targeted drugs can effectively overcome tumor drug resistance, making it the world's first epigenetic modulator approved for the treatment of solid tumors.
- In June 2021, it was approved in Japan as a monotherapy for the treatment of adult patients with relapsed or refractory (R/R) T-cell leukemia (ATL).
- In December 2021, Chidamide received its second approval in Japan—as a monotherapy for the treatment of patients with relapsed or refractory peripheral T-cell lymphoma.
- In March 2023, it was approved in Chinese Taiwan, for the treatment of postmenopausal patients with locally advanced or metastatic breast cancer that is hormone receptor-positive and human epidermal growth factor receptor 2 (HER2)-negative, and has relapsed or progressed after endocrine therapy (in combination with exemestane).
- In April 2024, it was approved in China in combination with R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone) for the treatment of treatment-naive patients with MYC- and BCL2-positive diffuse large B-cell lymphoma (DLBCL).
- In January 2025, the diffuse large B-cell lymphoma (DLBCL) indication of Chidamide was included in the NRDL for the first time, becoming the only oral innovative drug for first-line DLBCL treatment covered by medical insurance.
- In January 2026, Chidamide was officially included in the National Reimbursement Drug List (NRDL) under routine Category B;
Global Pipeline of Investigational Indications:
- A Phase III clinical study of Chidamide in combination with sintilimab and bevacizumab for the treatment of advanced microsatellite stable or mismatch repair proficient (MSS/pMMR) colorectal cancer patients who failed ≥2 lines of standard therapy is currently ongoing. Enrollment was completed by December 2025, and patients are currently under treatment follow-up.
- The randomized, double-blind, active-controlled Phase III clinical study of Chidamide in combination with nivolumab as first-line therapy for advanced melanoma, conducted by the company's international partner HUYABIO (HuaYa Bioscience International) across 17 countries worldwide, has completed enrollment, and patients are currently under treatment follow-up;
- A randomized, double-blind, placebo-controlled, multicenter Phase III clinical study of Chidamide in combination with CHOP for previously untreated peripheral T-cell lymphoma with follicular helper T-cell phenotype (PTCL-TFH) is currently ongoing;
- NW001 (an epigenetic immune ADC drug) is in the late preclinical research stage;
- In addition to the above, the company is conducting multiple investigator-initiated clinical studies of Chidamide as monotherapy or in combination with immunotherapy or other drugs for hematologic malignancies, solid tumors, and other indications.

